Question
- A ⟨100⟩ silicon wafer is oxidised in dry O₂ at 1000°C for 2 hours. Calculate the oxide thickness grown. Use Table 1 and the Deal-Grove model.
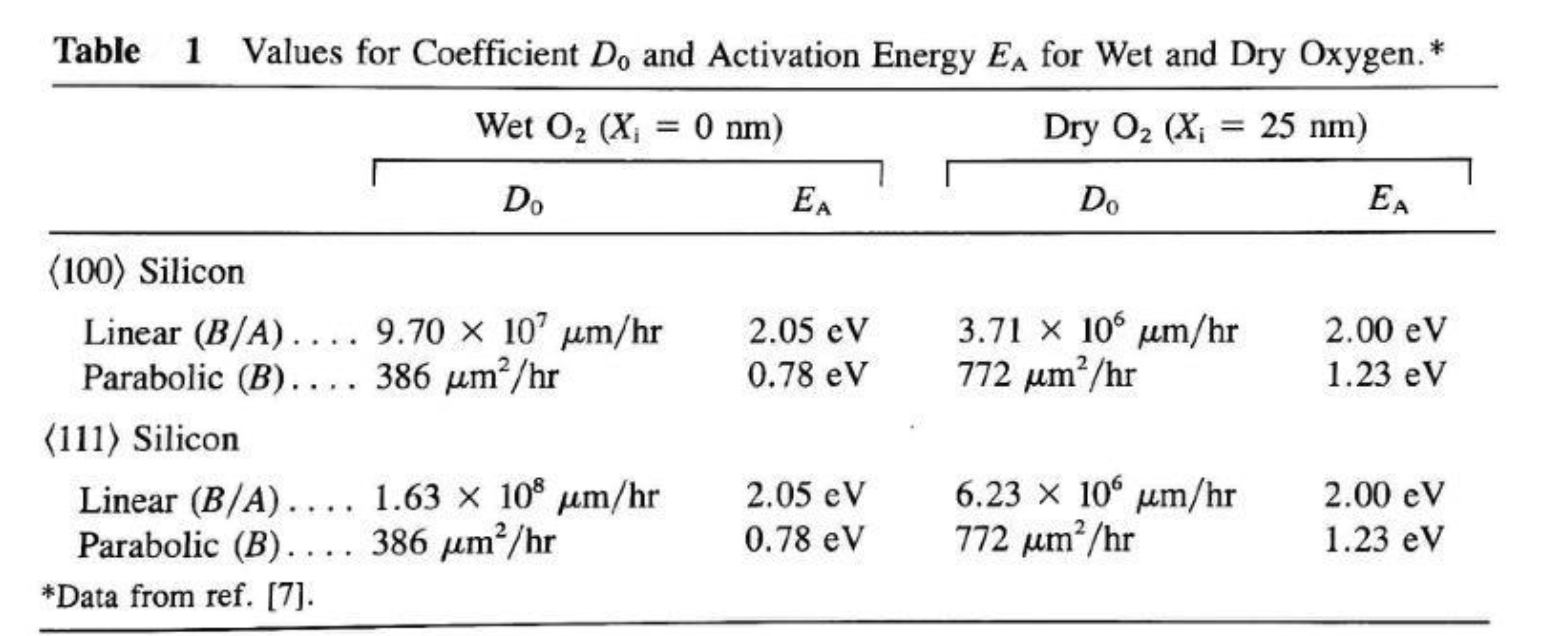
Step 1: Extract A and B at 1000°C dry, ⟨100⟩
From Table 1, dry O₂, ⟨100⟩ (X_i = 25 nm initial oxide assumed):
- B/A: D₀ = 3.71×10⁶ μm/hr, E_A = 2.00 eV
- B: D₀ = 772 μm²/hr, E_A = 1.23 eV
At T = 1273 K, k_BT = 0.1097 eV:
Step 2: Account for initial oxide X_i = 25 nm = 0.025 μm
Step 3: Solve for X at t = 2 hr
Quadratic in X:
2. A ⟨111⟩ silicon wafer already has 150 nm of SiO₂ from a previous process. It is placed in a wet O₂ furnace at 1200°C. How long to grow a further 400 nm?
Step 1: A and B at 1200°C wet, ⟨111⟩
At T = 1473 K, k_BT = (8.617×10⁻⁵)(1473) = 0.1269 eV
Step 2: τ for X_i = 0.150 μm
Step 3: t + τ for X_f = 0.550 μm
High temperature wet oxidation is fast — makes sense physically.
3. A process requires exactly 500 nm of gate oxide on ⟨100⟩ silicon. The engineer proposes a two-step process: first dry O₂ at 1100°C, then wet O₂ at 1000°C. The dry step runs for 3 hours. How long is the wet step?
Step 1: Oxide grown in dry step — A, B at 1100°C dry ⟨100⟩
T = 1373 K, k_BT = (8.617×10⁻⁵)(1373) = 0.1183 eV
Initial oxide X_i = 25 nm = 0.025 μm:
Solve for X after t = 3 hr:
Oxide after dry step: 211.5 nm
Step 2: Wet step at 1000°C ⟨100⟩ — A, B already calculated in Q6(c) above
A = 0.424 μm, B = 0.3142 μm²/hr
τ for X_i = 0.2115 μm:
t + τ for X_f = 0.500 μm:
4. Two identical ⟨100⟩ wafers start bare. Wafer 1 is oxidised in wet O₂ at 900°C for 10 hours. Wafer 2 is oxidised in dry O₂ at 1100°C for 10 hours. Which has more oxide, and by how much?
Wafer 1: wet O₂ at 900°C ⟨100⟩
T = 1173 K, k_BT = 0.1011 eV
X_i = 0 nm (bare), so τ = 0.
Wafer 2: dry O₂ at 1100°C — from Q3 above
A = 0.1403 μm, B = 0.02343 μm²/hr, τ = 0.1764 hr (for X_i = 25 nm)
Comparison:
| Wafer | Conditions | Oxide thickness |
|---|---|---|
| 1 | Wet 900°C, 10 hr | 860 nm |
| 2 | Dry 1100°C, 10 hr | 423 nm |
Wafer 1 has ~437 nm more oxide — despite being at 300°C lower temperature. This illustrates that wet oxidation is dramatically faster than dry due to the much higher diffusivity and solubility of H₂O in SiO₂ compared to O₂, which dominates over the temperature disadvantage.