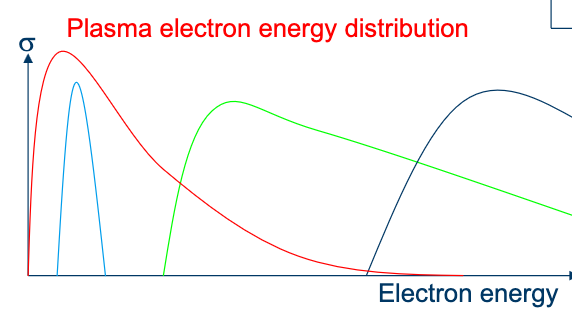
 The peak being , referring to the electron temperature
The dominant effect is not the voltage but pressure, which forces collisons to happen more frequently.
The peak being , referring to the electron temperature
The dominant effect is not the voltage but pressure, which forces collisons to happen more frequently.
A 1eV electron has velocity where and , so
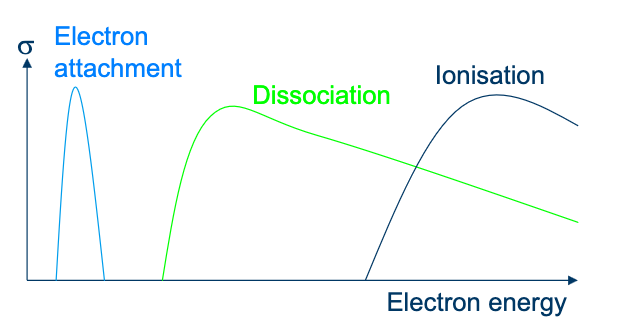
Dissociation
Leading to Radicals
Radical burns polymeric materials. Radical F attacks many materials - and is a gas.
Ionisation
An energetic electron can knock an electron out of an atom or molecule, leaving a positively charged ion. Notice that one electron goes into the reaction and two come out. This process therefore helps to sustain the plasma, creating a positive species and more negative ones.
Also: Dissociation and ionisation can and do often occur together.
Excitation
Here the atom or molecule, following the collision with the electron, is in an excited state. The atom or molecule will revert to the ground state and a photon of light of frequency is emitted. For example: + light These interactions are not thought to play a strong role in etching. The light given off however is very useful in understanding what species are in the plasma and is widely used in end-point detection. An optical spectrometer is a part of an etching laboratory. The excited state may be an electronic on or it may be a vibration mode.
Dissociative Electron Attachment
Some atoms and molecules gain energy by capturing an electron - they are said to have an electron affinity. The ions that are formed have a negative charge.
For example:
SF6 is used in waveguides to avoid arcing as it cleans up electrons. It is also a difficult gas in which to maintain a weak discharge. As we will learn later negative ions do not normally etch as the driven electrode is negatively charged.
The energy acquired by the molecule in the capture of an electron can cause the molecule to dissociate. For example: Note that one electron goes in and none comes out. It is more difficult to sustain a plasma when the gas has electron affinity.