Fick’s first law relates the diffusive flux to the gradient of the concentration. It postulates that the flux goes from regions of high concentration to regions of low concentration, with a magnitude that is proportional to the concentration gradient (spatial derivative), or in simplistic terms the concept that a solute will move from a region of high concentration to a region of low concentration across a concentration gradient.
- is the diffusion flux and measures the amount of substance that will flow through a unit area during a unit time interval.
- is the diffusion coefficient or diffusivity.
- is the concentration gradient
Broadly we can then model D across Temperature (T) as:
- is the diffusion coefficient, as before.
- is the pre-exponential factor / frequency factor — the theoretical maximum diffusivity at infinite temperature.
- is the activation energy — the energy barrier a particle must overcome to make a diffusive jump. or
- is the Boltzmann constant. (or )
- is the absolute temperature.
Typically we plot this relation for different chemicals as an Arrhenius Plot:
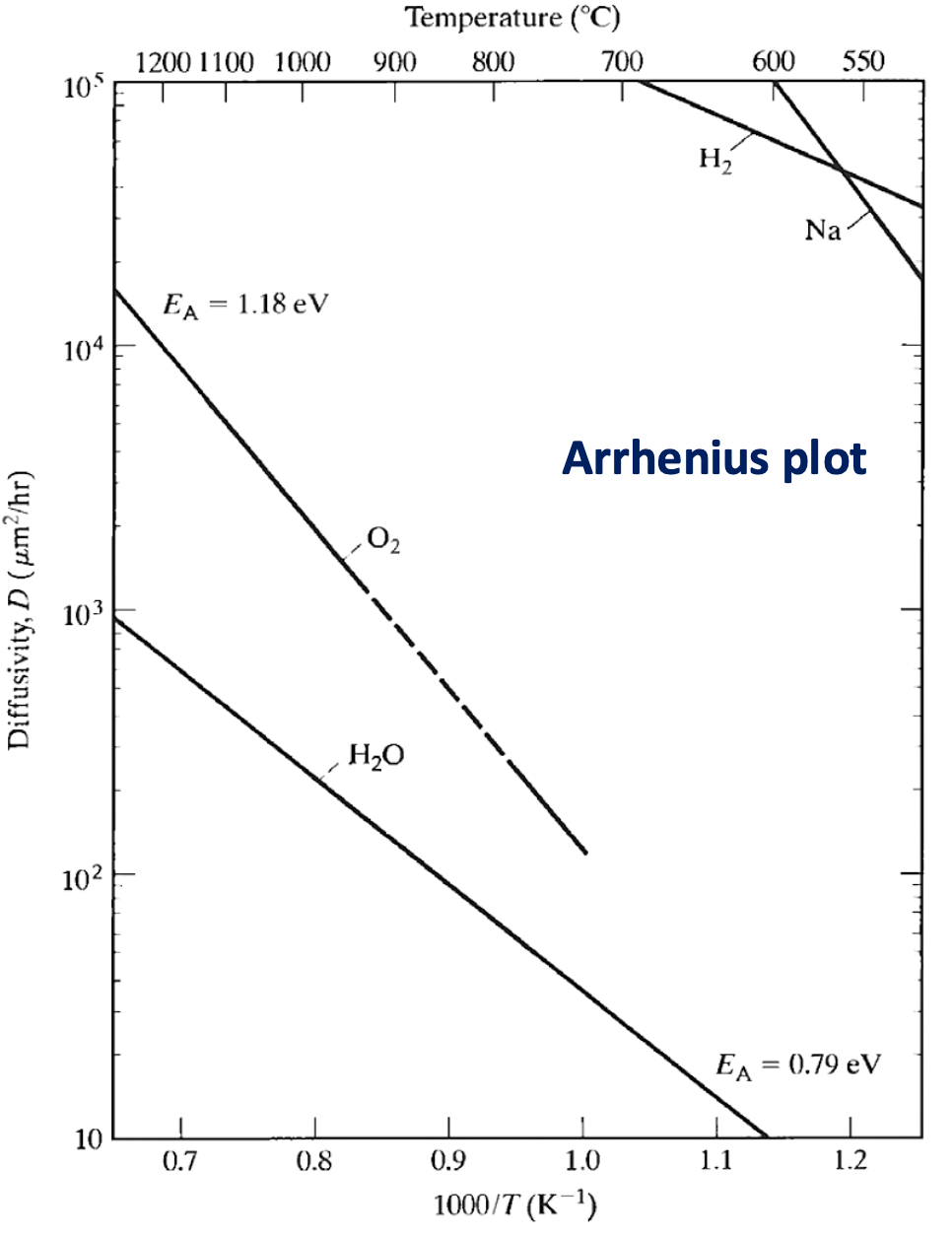
For a single rate-limited thermally activated process, an Arrhenius plot gives a straight line, from which the activation energy () and the pre-exponential factor can both be determined.