Many ways to grow Oxide Films:
Thermal Oxidation is generally the best for producing the least defects
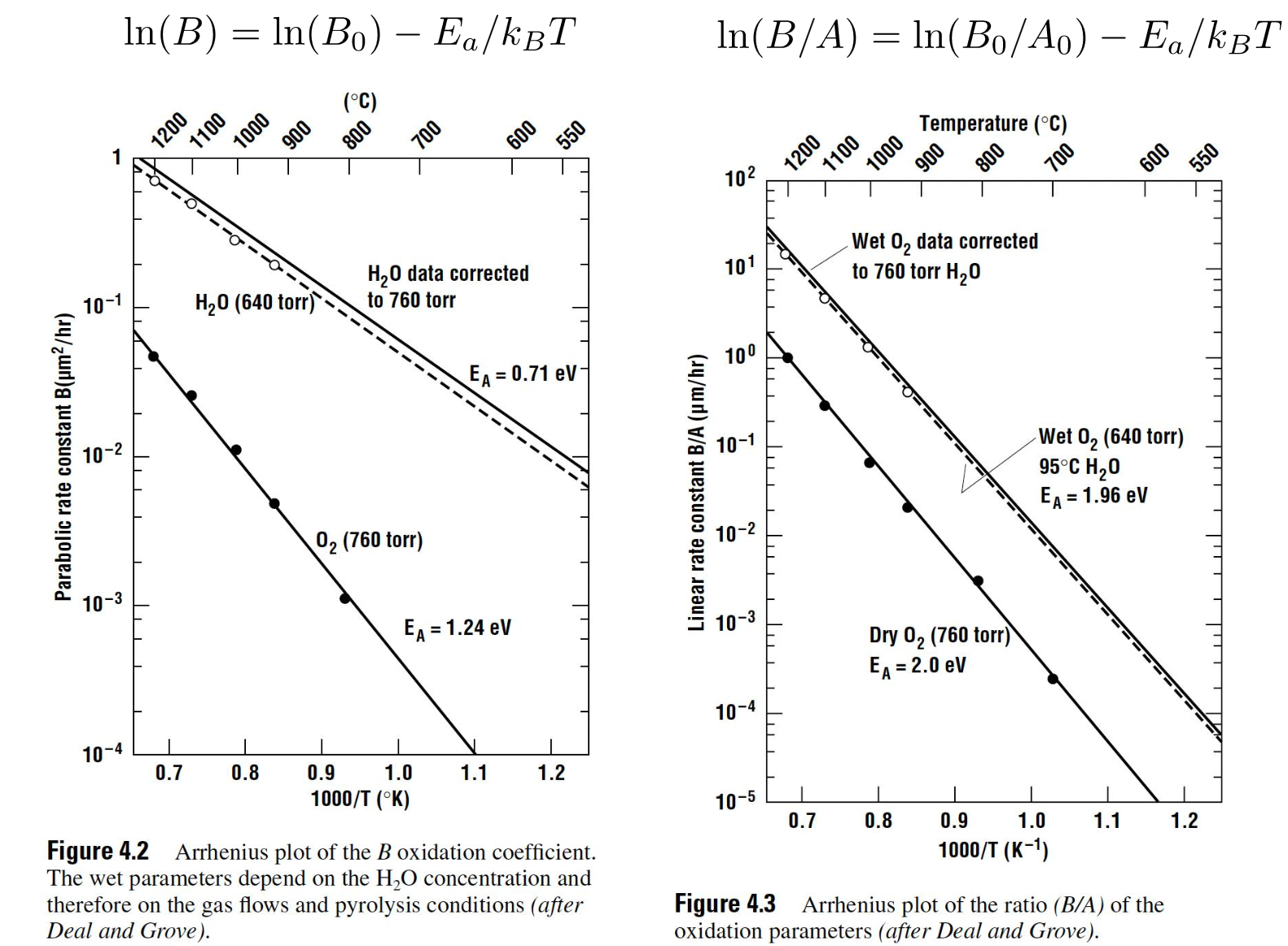
The diffusion is modelled by Fick’s Law We can also use the Deal-Grove Model to predict oxidation rates expect for thin oxides in pure
Factors Affecting Oxidation
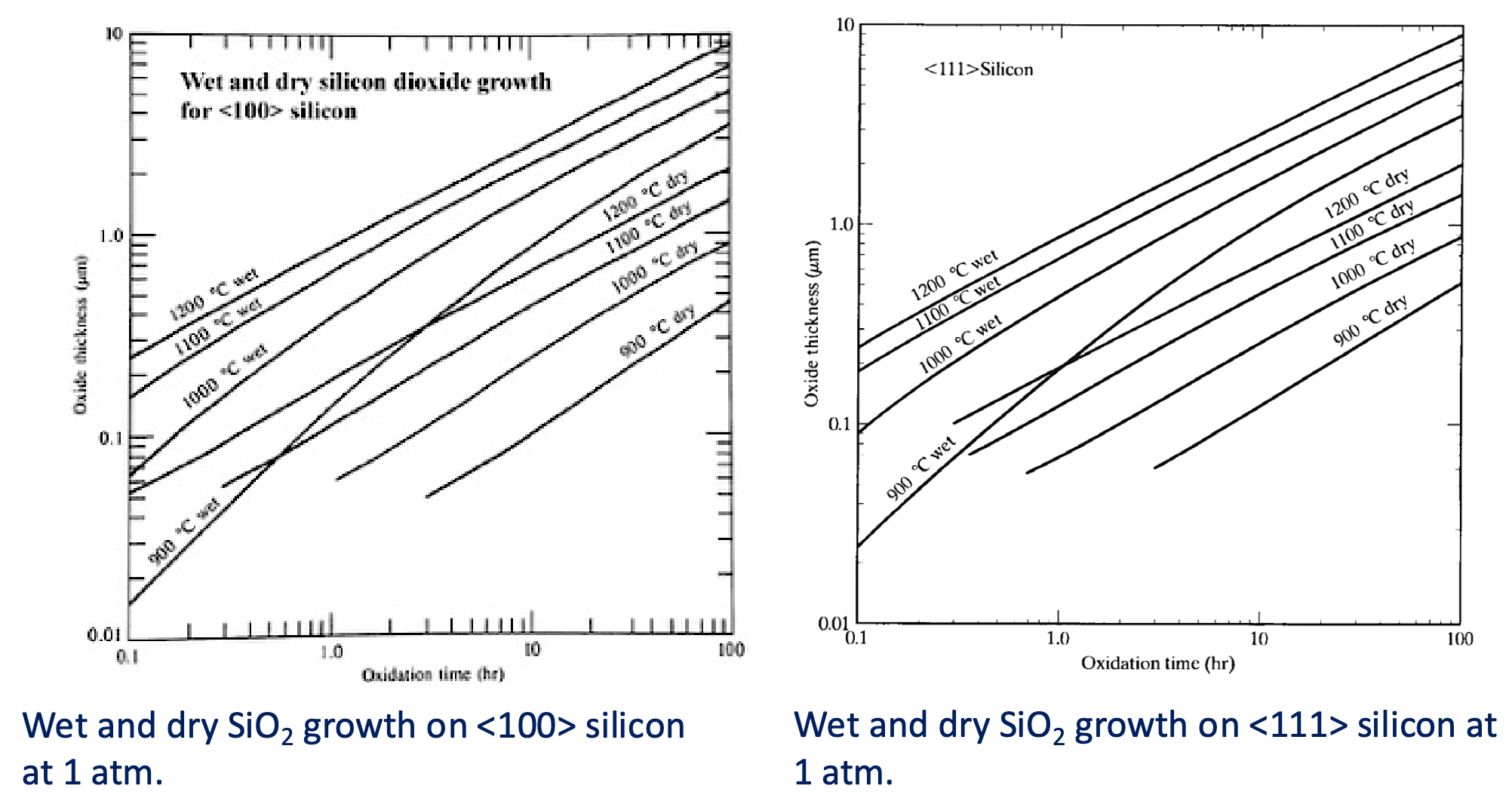
Wet and Dry Oxidation
Water vapor has a much higher solubility than oxygen in , which accounts for the much higher oxide growth rate in a wet atmosphere.
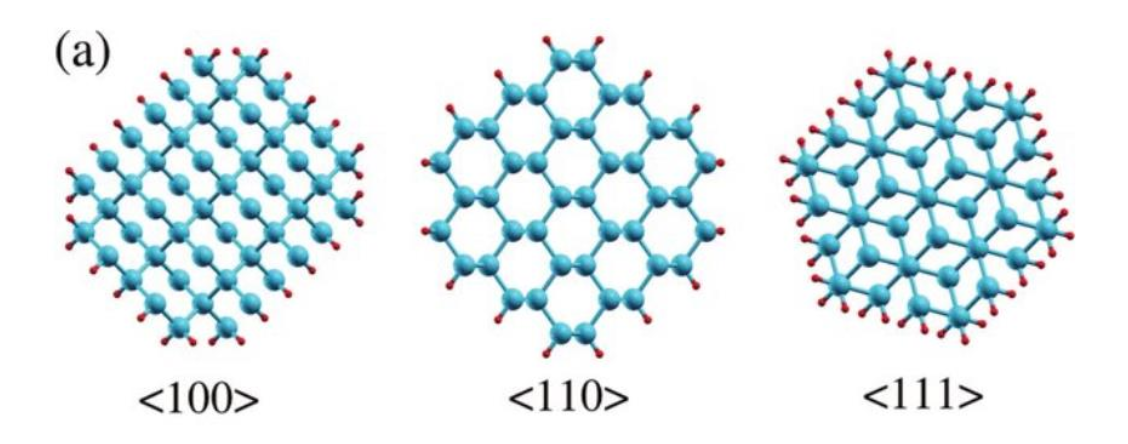
Crystal Orientation
The oxidation rate is dependent on the number of available reaction sites on the silicon substrate.
Oxidation on the <111> crystal plane occurs at a higher rate because there are a higher number of surface atoms/chemical bonds than the <100> crystal plane.
- Most IC is made of <100> silicon.
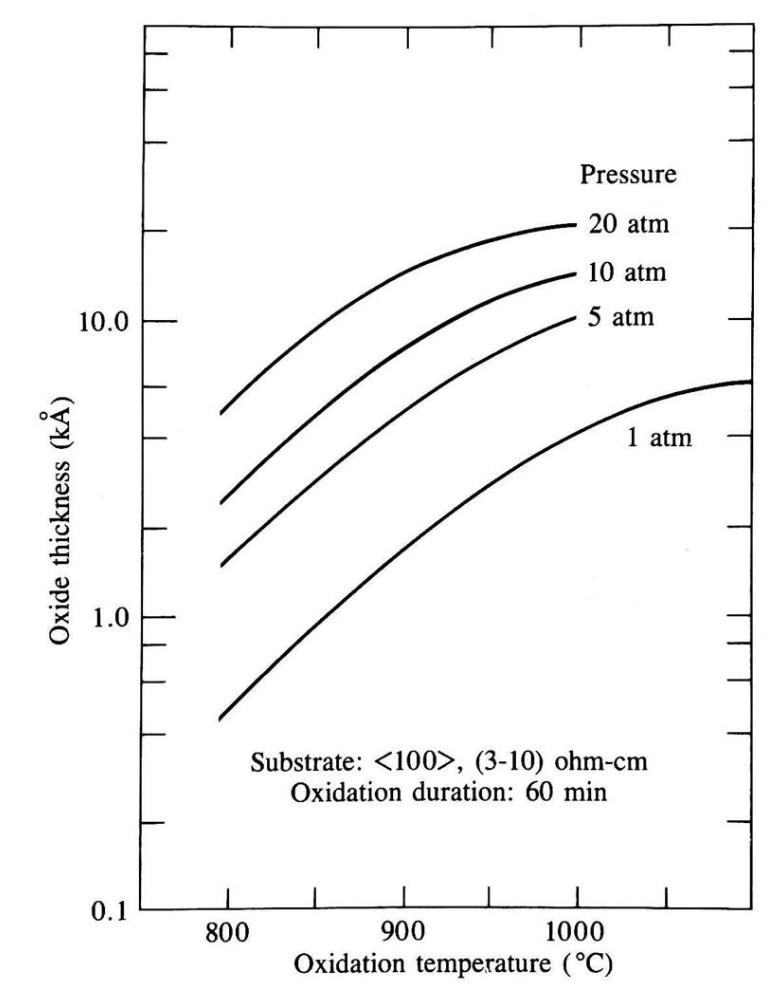
Pressure
Impurity Doping
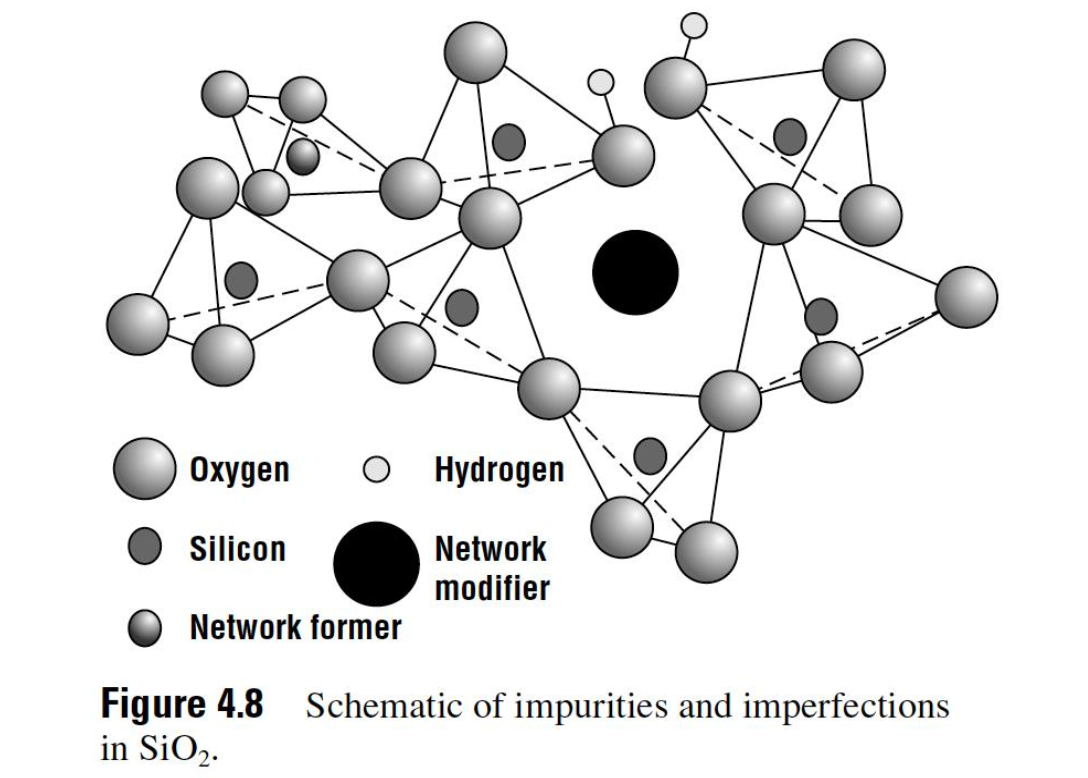
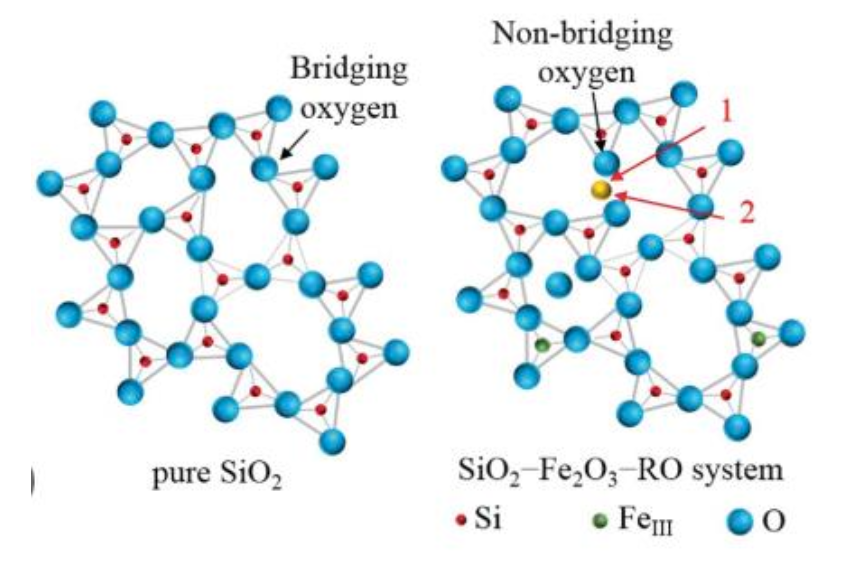
Bridging vs Non-Bridging Oxygen
In fused silica some oxygen atoms, called bridging oxygen sites, are bonded to
two silicon atoms. Some oxygen atoms are nonbridging, bonded to only one
silicon. Thermally grown SiO2 then can be considered to consist primarily of a
randomly oriented network of polyhedra. The larger the fraction of bridging to
nonbridging sites, the most cohesive and less prone to damage the oxide is.
Dry oxides have a much larger ratio of bridging to nonbridging sites compared
to wet oxides.
A variety of impurities can also exist in thermal oxides. Some of the most common are water-related complexes. If is present during the growth, one reaction that can occur is the reduction of a bridging oxygen site into two hydroxyls:
These hydrogen atoms are only weakly bonded and can be removed under electrical stress or ionizing radiation, leaving a trap or potential charged state in the oxide.
Other impurities are intentionally incorporated into thermally deposited SiO2 to change its physical and electrical properties. Substitutional impurities that replace the silicon atom are called network formers. The most common network formers are boron and phosphorus. These impurities tend to reduce the bridging-to-nonbridging ratio, which allows the glass to flow at lower temperature. These impurities, however, are normally used in deposited oxides rather than in thermal oxides.