Absorption
Photons at a energy equal to or above the band gap of a semiconductor can cause an electron in the valence band to be promoted to the conduction band:
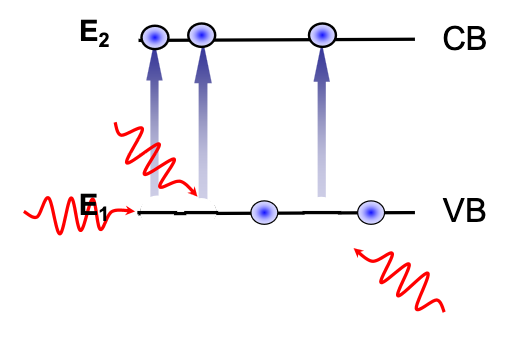
This is the principle of photodetectors.
Spontaneous Emission
Electrons in the Conduction band can spontaneously lose energy through releasing a photon, and fall to the Valence band:
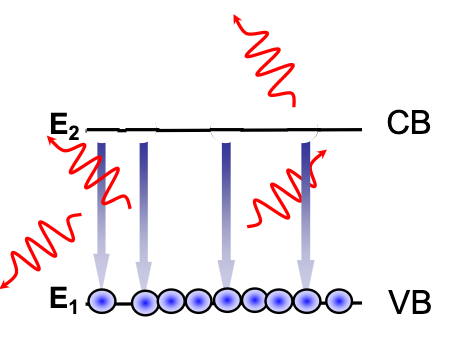
This is the principle for LEDs, whats important to note is that for a P-N junction being driven under forward bias is not in equilibrium, increasing the likelihood of electrons releasing a photon to try balance this:
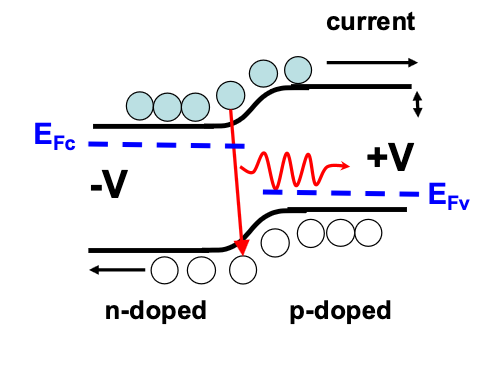
Stimulated Emission
For a high concentration of electrons in the conduction band and holes in the valence band, incoming photons can stimulate the production of another identical photon (energy and phase).
This is the principle of a Semiconductor Optical Amplifier and therefore Light Amplified by Stimulated Emission of Radition (LASER), where you essentially keep feeding back photons into a system that uses Stimulated Emission to make more photons, these constructively intefere at wavelengths according to the size of the cavity.
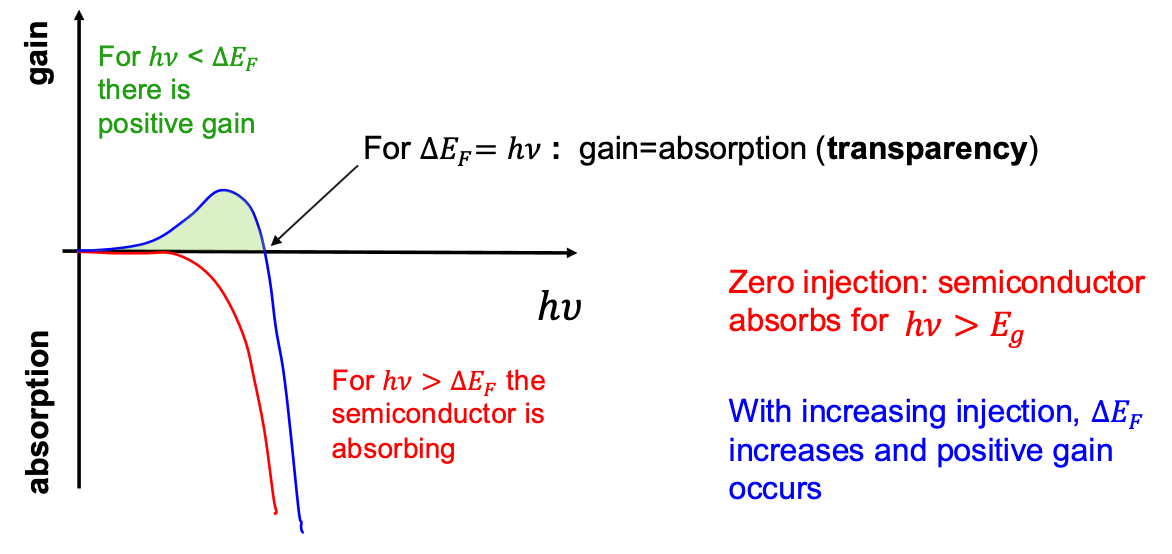
Gain and Absorption Spectra
Equilibrium without Injection:
At low injection: therefore spontaneous emission dominates This is the basis for a LED At higher injection: This is the basis for optical gain
is the Fermi Energy Splitting, in this case it relates to the driving voltage. the photon energy.
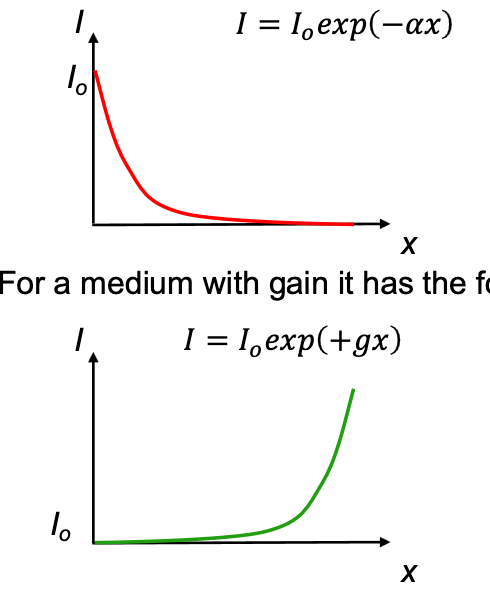
Beer-Lambert Law
How much the intensity of a field varies as a function of distance in an absorbing medium