If we fire a laser with a photon energy larger than the band gap energy, it will excite electrons from the valence band to the conduction band, and leave a hole behind. These will then proceed to lose energy via phonons, until the finally recombine, producing a photon:
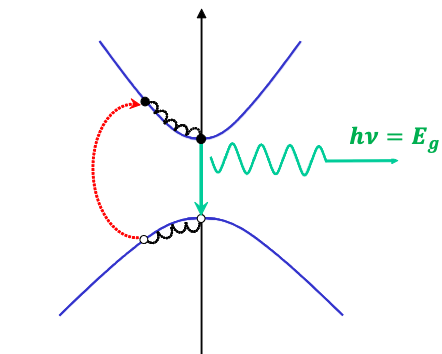
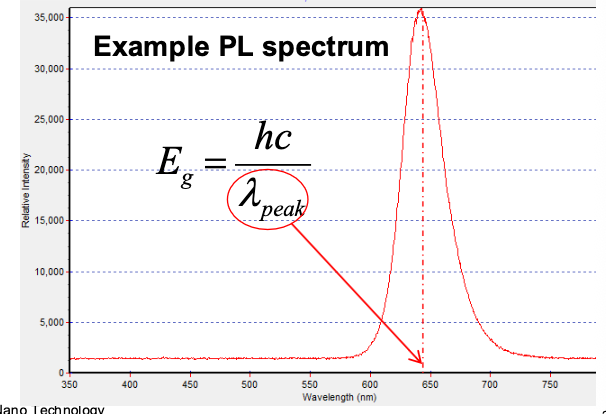
This actually leads to a slightly wider emission spectrum due to thermal energy, but the peak wavelength should be the band gap:
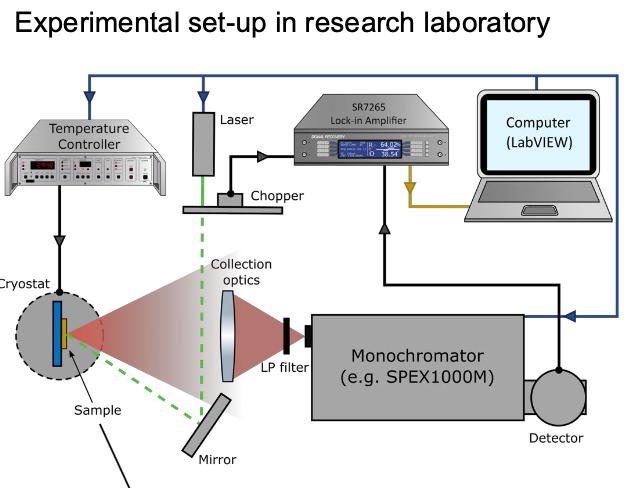
Setup
What can we learn from Photoluminescence
The peak wavelength provides an indication of the band gap, and hence the Sb composition
The peak intensity provides an indication of how good the material is at emitting light. It is a measure of optical quality
The spectral width (linewidth), usually Full Width at Half Maximum (FWHM), is a measure of material homogeneity / uniformity