Thin Films Tutorial Questions
Question
- Mention at least 4 factors that influence the growth of SiO₂ during thermal oxidation of silicon.
Answer
(1) Temperature — higher T increases diffusion and reaction rates exponentially (Arrhenius). (2) Oxidant — wet oxidation (H₂O) is faster than dry (O₂) because water diffuses more readily through SiO₂. (3) Crystal orientation — (111) oxidises faster than (100) due to higher atom density at the surface. (4) Existing oxide thickness — as oxide grows, diffusion distance increases, slowing growth (parabolic regime). Also: pressure, doping level (heavily doped Si oxidises faster), and chlorine additives.
Question
- What does ‘wet oxidation’ refer to in SiO₂ thin films? A) Hydrating the SiO₂ surface for better adhesion B) Oxidation of silicon in a water vapour environment to form SiO₂ C) Application of liquid coatings D) A cleaning process using water E) Reducing the dielectric constant
Answer
B Si + 2H₂O → SiO₂ + 2H₂. Faster than dry oxidation (Si + O₂ → SiO₂) because H₂O diffuses more readily through existing oxide, but produces lower quality (less dense) oxide.
Question
- For SiO₂ thin films, what effect does increasing deposition temperature have on density? A) Increases the density B) Decreases the density C) No effect D) First increases, then decreases E) Varies randomly
Answer
A Higher temperature gives atoms more energy to rearrange into a denser, more ordered structure. Thermal oxides grown at higher temperatures are closer to the theoretical density of fused silica.
Question
- What role does thickness of SiO₂ thin films play in optical applications? A) Determines the colour B) Affects the diffraction pattern C) Controls the refractive index D) Alters the absorption spectrum E) Influences the interference colour
Answer
E Thin film interference produces constructive/destructive interference depending on the optical path length (2nt). Different thicknesses produce different interference colours — this is why oxide-covered silicon wafers show characteristic colours that indicate thickness.
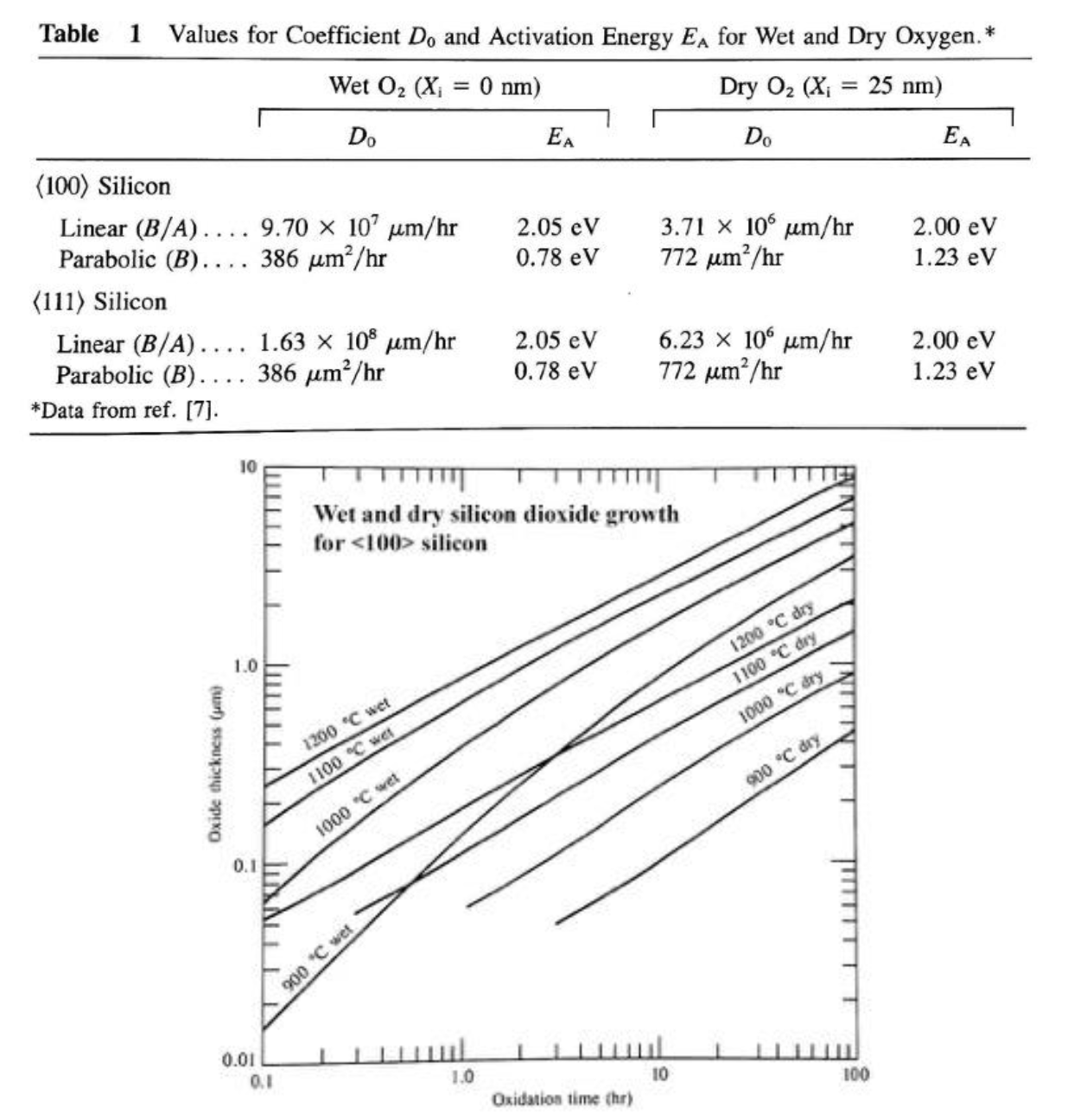
Question
- (a) Time to grow 200 nm of SiO₂ on ⟨100⟩ Si at 1100°C in dry oxygen?
Answer
From the graph, the 1100°C dry curve reaches 0.2 µm at approximately 1.5-2 hours.
Question
(b) Starting from 200 nm, grow an additional 300 nm (total 500 nm) in wet oxygen at 1000°C. How long?
Answer
On the 1000°C wet curve: 200 nm (0.2 µm) is reached at ~0.15 hr, and 500 nm (0.5 µm) at ~0.8 hr. The additional growth time is the difference: ~0.8 - 0.15 ≈ 0.65 hr ≈ 40 minutes. We subtract the time to reach the starting thickness because the existing 200nm oxide was already present.
Question
(c) Repeat part (b) using oxidation theory and Table 1.
Answer
Deal-Grove model: x² + Ax = B(t + τ), where A = 2D/k_s (linear coefficient) and B is the parabolic rate constant. At 1000°C wet, ⟨100⟩: B/A = 9.70×10⁷ × exp(-2.05/(kT)) and B = 386 × exp(-0.78/(kT)) kT at 1000°C (1273 K) = 8.617×10⁻⁵ × 1273 = 0.1097 eV B/A = 9.70×10⁷ × exp(-2.05/0.1097) = 9.70×10⁷ × exp(-18.69) = 9.70×10⁷ × 7.60×10⁻⁹ = 0.737 µm/hr B = 386 × exp(-0.78/0.1097) = 386 × exp(-7.11) = 386 × 8.14×10⁻⁴ = 0.314 µm²/hr A = B/(B/A) = 0.314/0.737 = 0.426 µm Find τ for existing 200nm oxide: (0.2)² + 0.426(0.2) = 0.314(t₁ + τ) → 0.04 + 0.0852 = 0.314(t₁ + τ) → t₁ + τ = 0.399 hr For total 500nm: (0.5)² + 0.426(0.5) = 0.314(t₂ + τ) → 0.25 + 0.213 = 0.314(t₂ + τ) → t₂ + τ = 1.475 hr Additional time = (t₂ + τ) - (t₁ + τ) = 1.475 - 0.399 = 1.08 hr ≈ 65 minutes. (Slight difference from graphical estimate due to reading precision.)